Журнал "Гастроэнтерология" Том 56, №4, 2022
Вернуться к номеру
Кишковий фіброгенез при запальних захворюваннях кишечника
Авторы: Жайворонок М.Н. (1, 2), Залеський В.М. (3)
(1) — Національний університет охорони здоров’я України імені П.Л. Шупика, м. Київ, Україна
(2) — Медичне науково-практичне об’єднання «МедБуд», м. Київ, Україна
(3) — ДУ «Національний науковий центр «Інститут кардіології імені академіка М.Д.Стражеска» НАМН України», м. Київ, Україна
Рубрики: Гастроэнтерология
Разделы: Справочник специалиста
Версия для печати
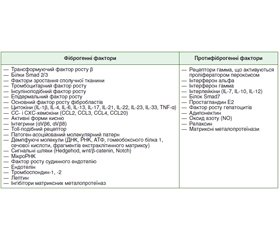
Стаття присвячена хронічним запальним процесам кишечника і їх ускладненням, що сприяють поступовому накопиченню глибоких трансмуральних уражень стінки кишечника, в тому числі звужень, розвитку непрохідності, абсцесів і нориць. Як запальні захворювання кишечника, так і їх хронічні ускладнення призводять до появи у хворих діареї, болю в животі, анемії, що пов’язана з кишковою патологією. Визначення активності захворювання й вираженості ускладнень має вирішальне значення в інтенсивності лікування на ранніх і наступних стадіях захворювання і моніторингу ефективності лікувальних заходів. В огляді розглянуті основні молекулярні медіатори фіброгенезу, підбиті підсумки розробки й розвитку технологій для візуалізації кишкового фіброзу, а також обговорюються можливості розширення кількісної магнітно-резонансної томографії, комп’ютерної томографії, ультразвукового дослідження й обнадійливий потенціал методів неінвазивної еластографії.
The article deals with chronic inflammatory processes of the intestines and their complications that contribute to the gradual accumulation of deep transmural lesions of the intestinal wall, including narrowings, development of obstruction, abscesses, and fistulas. Both inflammatory bowel diseases and their chronic complications lead to the onset of diarrhea, abdominal pain, anemia caused by intestinal pathology. Detecting the disease activity and complications severity is of crucial importance in the treatment intensity at early and later stages of the illness and when monitoring treatment measures effectiveness. The main molecular mediators of fibrogenesis are studied in the article, the results of development of intestinal fibrosis visualization technologies are summarized, possibilities for expanding the quantitative magnetic resonance imaging, computed tomography, ultrasound and encouraging potential of non-invasive elastography methods are discussed.
кишковий фіброз; хвороба Крона; виразковий коліт; молекулярні механізми; біомаркери; візуалізація; магнітно-резонансна томографія; комп’ютерна томографія; ультразвукове дослідження; еластографія
intestinal fibrosis; Crohn’s disease; ulcerative colitis; molecular mechanisms; biomarkers; imaging methods; magnetic resonance imaging; computed tomography; ultrasound; elastography
Для ознакомления с полным содержанием статьи необходимо оформить подписку на журнал.
- Latella G., Rogler G., Bamias G. et al. Results of the 4th scientific workshop of the ECCO (I): pathophysiology of intestinal fibrosis in IBD. J Crohns Colitis. 2014 Oct. 8(10). 1147-65.
- Richter K., Konzack A., Pihlajaniemi T., Heljasvaara R., Kietzmann T. Redox-fibrosis: Impact of TGFβ1 on ROS generators, mediators and functional consequences. Redox Biol. 2015 Dec. 6. 344-352.
- Rockey D.C., Bell P.D., Hill J.A. Fibrosis — A Common Pathway to Organ Injury and Failure. N Engl J Med. 2015 Mar 19. 372(12). 1138-49.
- Li S., Lu R., Shu L. et al. An integrated map of fibroblastic populations in human colon mucosa and cancer tissues. Commun Biol. 2022 Dec 3. 5(1). 1326. doi: 10.1038/s42003-022-04298-5.
- Orenstein J.M. An ultrastructural pathologist’s views on fibroblasts, modified smooth muscle cells, wound healing, stenosing arteriopathies, Kawasaki disease, Dupuytren’s contracture, and the stroma of carcinomas. Ultrastruct Pathol. 2020 Jan 2. 44(1). 2-14. doi: 10.1080/01913123.2019.1704332.
- Tobiume M., Mitsuhashi A., Saijo A. et al. Analysis of the chemotactic factors for tumor-infiltrating fibrocytes and their prognostic significances in lung cancer. Oncol Lett. 2022 Sep 30. 24(5). 417. doi: 10.3892/ol.2022.13537.
- She Z.G., Chang Y., Pang H.B., Han W., Chen H.Z., Smith J.W., Stallcup W.B. NG2 Proteoglycan Ablation Reduces Foam Cell Formation and Atherogenesis via Decreased Low-Density Lipoprotein Retention by Synthetic Smooth Muscle Cells. Arterioscler Thromb Vasc Biol. 2016 Jan. 36(1). 49-59. doi: 10.1161/ATVBAHA.115.306074.
- Šimková A., Bušek P., Šedo A., Konvalinka J. Molecular recognition of fibroblast activation protein for diagnostic and therapeutic applications. Biochim Biophys Acta Proteins Proteom. 2020 Jul. 1868(7). 140409. doi: 10.1016/j.bbapap.2020.140409.
- Nikoloudaki G. Functions of Matricellular Proteins in Dental Tissues and Their Emerging Roles in Orofacial Tissue Development, Maintenance, and Disease. Int J Mol Sci. 2021 Jun 21. 22(12). 6626. doi: 10.3390/ijms22126626.
- Huang Z., Zhang Z., Zhou C., Liu L., Huang C. Epithelial-mesenchymal transition: The history, regulatory mechanism, and cancer therapeutic opportunities. MedComm. 2022 May 18. 3(2). e144. doi: 10.1002/mco2.144
- Wynn T.A., Ramalingam T.R. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012 Jul 6. 18(7). 1028-40.
- Rieder F., Karrasch T., Ben-Horin S. et al. Results of the 2nd scientific workshop of the ECCO (III): basic mechanisms of intestinal healing. J Crohns Colitis. 2012 Apr. 6(3). 373-85.
- Matsuda M., Seki E. The liver fibrosis niche: Novel insights into the interplay between fibrosis-composing mesenchymal cells, immune cells, endothelial cells, and extracellular matrix. Food Chem Toxicol. 2020 Sep. 143. 111556. doi: 10.1016/j.fct.2020.111556.
- Latella G., Sferra R., Speca S., Vetuschi A., Gaudio E. Can we prevent, reduce or reverse intestinal fibrosis in IBD? Eur Rev Med Pharmacol Sci. 2013 May. 17(10). 1283-304.
- Valgio A. Systemic Fibroinflammatory Disorders. Springer Verlag, 2017. 243 p.
- Pottier N., Cauffiez C., Perrais M., Barbly P., Mari B. FibromiRs: translating molecular discoveries into new anti-fibrotic drugs. Trends Pharmacol Sci. 2014 Mar. 35(3). 119-26.
- Richter K., Kietzmann T. Reactive oxygen species and fibrosis: further evidence of a significant liaison. Cell Tissue Res. 2016 Sep. 365(3). 591-605.
- Ghatak S., Hascall V.C., Markwald R.R., Feghali-Bostwick C., Artlett C.M., Gooz M. et al. Transforming growth factor β1 (TGFβ1)-induced CD44V6-NOX4 signaling in pathogenesis of idiopathic pulmonary fibrosis. J Biol Chem. 2017 Jun 23. 292(25). 10490-10519. doi: 10.1074/jbc.M116.752469.
- Miao R., Wang L., Chen Z. et al. Advances in the study of nicotinamide adenine dinucleotide phosphate oxidase in myocardial remodeling. Front Cardiovasc Med. 2022 Nov 3. 9. 1000578. doi: 10.3389/fcvm.2022.1000578.
- Sedeek M., Nasrallah R., Touyz R.M., Hébert R.L. NADPH oxidases, reactive oxygen species, and the kidney: friend and foe. J Am Soc Nephrol. 2013 Oct. 24(10). 1512-8.
- Estornut C., Milara J., Bayarri M.A., Belhadj N., Cortijo J. Targeting Oxidative Stress as a Therapeutic Approach for Idiopathic Pulmonary Fibrosis. Front Pharmacol. 2022 Jan 21. 12. 794997. doi: 10.3389/fphar.2021.794997.
- Mortezaee K. Nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX) and liver fibrosis: A review. Cell Biochem Funct. 2018 Aug. 36(6). 292-302. doi: 10.1002/cbf.3351.
- Paik Y.H., Kim J., Aoyama T., De Minicis S., Bataller R., Brenner D.A. Role of NADPH oxidases in liver fibrosis. Antioxid Redox Signal. 2014 Jun 10. 20(17). 2854-72.
- Bernard K., Hecker L., Luckhardt T.R., Cheng G., Thannickal V.J. NADPH oxidases in lung health and disease. Antioxid Redox Signal. 2014 Jun 10. 20(17). 2838-53.
- Papaetis G.S. Pioglitazone in diabetic kidney disease: forgotten but not gone. Arch Med Sci Atheroscler Dis. 2022 Aug 8. 7. e78-e93. doi: 10.5114/amsad/151046. PMID: 36158067; PMCID: PMC9487837.
- Cemma M., Grinstein S., Brumell J.H. Autophagy proteins are not universally required for phagosome maturation. Autophagy. 2016 Sep. 12(9). 1440-6. doi: 10.1080/15548627.2016.1191724.
- Radwan M.I., Pasha H.F., Mohamed R.H., Hussien H.I., El-Khshab M.N. Influence of transforming growth factor-β1 and tumor necrosis factor-α genes polymorphisms on the development of cirrhosis and hepatocellular carcinoma in chronic hepatitis C patients. Cytokine. 2012 Oct. 60(1). 271-6.
- Manickam N., Patel M., Griendling K.K., Gorin Y., Bar–nes J.L. RhoA/Rho kinase mediates TGF-β1-induced kidney myofibroblast activation through Poldip2/Nox4-derived reactive oxygen species. Am J Physiol Renal Physiol. 2014 Jul 15. 307(2). F159-71. doi: 10.1152/ajprenal.00546.2013.
- Boudreau H.E., Casterline B.W., Rada B., Korzeniowska A., Leto T.L. Nox4 involvement in TGF-beta and SMAD3-driven induction of the epithelial-to-mesenchymal transition and migration of breast epithelial cells. Free Radic Biol Med. 2012 Oct 1. 53(7). 1489-99.
- Roodnat A.W., Callaghan B., Doyle C., Henry M., Goljanek-Whysall K., Simpson D.A. Genome-Wide RNA Sequencing of Human Trabecular Meshwork Cells Treated with TGF-β1: Relevance to Pseudoexfoliation Glaucoma. Biomolecules. 2022 Nov 15. 12(11). 1693. doi: 10.3390/biom12111693.
- Sato K., Hirano I., Sekine H. et al. An immortalized cell line derived from renal erythropoietin-producing (REP) cells demonstrates their potential to transform into myofibroblasts. Sci Rep. 2019 Aug 2. 9(1). 11254. doi: 10.1038/s41598-019-47766-5. PMID: 31375751; PMCID: PMC6677766.
- Xu X., Tan X., Tampe B., Sanchez E., Zeisberg M., Zeisberg E.M. Snail Is a Direct Target of Hypoxia-inducible Factor 1α (HIF1α) in Hypoxia-induced Endothelial to Mesenchymal Transition of Human Coronary Endothelial Biol Chem. 2015 Jul 3. 290(27). 16653-64.
- Huang P., Gu X.J., Huang M.Y., Tan J.H., Wang J. Down-regulation of LINC00667 hinders renal tubular epithelial cell apoptosis and fibrosis through miR-34c. Clin Transl Oncol. 2021 Mar. 23(3). 572-581. doi: 10.1007/s12094-020-02451-2.
- Chen S.L., Zheng M.H., Shi K.Q., Yang T., Chen Y.P. A new strategy for treatment of liver fibrosis: letting MicroRNAs do the job. BioDrugs. 2013 Feb. 27(1). 25-34.
- Cui R.R., Li S.J., Liu L.J. et al. MicroRNA-204 regulates vascular smooth muscle cell calcification in vitro and in vivo. Cardiovasc Res. 2012 Nov 1. 96(2). 320-9.
- Nagao M., Lyu Q., Zhao Q., et al. Coronary Disease-Associa–ted Gene TCF21 Inhibits Smooth Muscle Cell Differentiation by Bloc–king the Myocardin-Serum Response Factor Pathway. Circ Res. 2020 Feb 14. 126(4). 517-529. doi: 10.1161/CIRCRESAHA.119.315968.
- Nijhuis A., Biancheri P., Lewis A. et al. A In Crohn’s disease fibrosis-reduced expression of the miR-29 family enhances collagen expression in intestinal fibroblasts. Clin Sci (Lond). 2014 Sep. 127(5). 341-50.
- Bian E.B., Li J., Zhao B. miR-29, a potential therapeutic target for liver fibrosis. Gene. 2014 Jul 10. 544(2). 259-60.
- Chen J., Yu Y., Li S. et al. MicroRNA-30a ameliorates hepatic fibrosis by inhibiting Beclin1-mediated autophagy. J Cell Mol Med. 2017 Dec. 21(12). 3679-3692. doi: 10.1111/jcmm.13278.
- Irungbam K., Roderfeld M., Glimm H. et al. Cholestasis impairs hepatic lipid storage via AMPK and CREB signaling in hepatitis B virus surface protein transgenic mice. Lab Invest. 2020 Nov. 100(11). 1411-1424. doi: 10.1038/s41374-020-0457-9.
- Yu X., Elfimova N., Müller M. et al. Autophagy-Related Activation of Hepatic Stellate Cells Reduces Cellular miR-29a by Promoting Its Vesicular Secretion. Cell Mol Gastroenterol Hepatol. 2022. 13(6). 1701-1716. doi: 10.1016/j.jcmgh.2022.02.013.
- Bhattacharyya S., Kelley K., Melichian D.S., Tamaki Z., Fang F., Su Y., Feng G. Toll-like receptor 4 signaling augments transforming growth factor-β responses: a novel mechanism for maintaining and amplifying fibrosis in scleroderma. Am J Pathol. 2013 Jan. 182(1). 192-205.
- Feki S., Bouzid D., Abida O. et al. Genetic association and phenotypic correlation of TLR4 but not NOD2 variants with Tunisian inflammatory bowel disease. J Dig Dis. 2017 Nov. 18(11). 625-633. doi: 10.1111/1751-2980.12552.
- Yang J., Zhou C.Z., Zhu R., Fan H., Liu X.X., Duan X.Y., Tang Q., Shou Z.X., Zuo D.M. miR-200b-containing microvesicles attenuate experimental colitis associated intestinal fibrosis by inhibiting epithelial-mesenchymal transition. J Gastroenterol Hepatol. 2017 Dec. 32(12). 1966-1974. doi: 10.1111/jgh.13797.
- Radford S.J., Taylor S., Moran G. Ultrasound use to assess Crohn’s disease in the UK: a survey of British Society of Gastroentero–logy Inflammatory Bowel Disease Group members. Frontline Gastroente–rol. 2022 Jan 18. 13(6). 471-476. doi: 10.1136/flgastro-2021-102065.
- Pariente B., Mary J.Y., Danese S., Chowers Y., De Cruz P., D’Haens G., Loftus E.V. Jr. Development of the Lémann index to assess digestive tract damage in patients with Crohn’s disease. Gastroenterology. 2014 Feb. 146(2). 374-82.e1.
- Ordás I., Rimola J., Rodríguez S., Paredes J.M., Martínez-Pérez M.J., Blanc E., Arévalo J.A. Accuracy of magnetic resonance enterography in assessing response to therapy and mucosal healing in patients with Crohn’s disease. Gastroenterology. 2014 Feb. 146(2). 374-82.e1.
- Rimola J., Torres J., Kumar S., Taylor S.A., Kucharzik T. Recent advances in clinical practice: advances in cross-sectional imaging in inflammatory bowel disease. Gut. 2022 Dec. 71(12). 2587-2597. doi: 10.1136/gutjnl-2021-326562.
- Coelho R., Ribeiro H., Maconi G. Bowel Thickening in Crohn’s Disease: Fibrosis or Inflammation? Diagnostic Ultrasound Imaging Tools. Inflamm Bowel Dis. 2017 Jan. 23(1). 23-34. doi: 10.1097/MIB.0000000000000997.
- Nylund K., Jirik R., Mezl M., Leh S., Hausken T., Pfeffer F., Ødegaard S. Quantitative contrast-enhanced ultrasound comparison between inflammatory and fibrotic lesions in patients with Crohn’s di–sease. Ultrasound Med Biol. 2013 Jul. 39(7). 1197-206.
- Pescatori L.C., Mauri G., Savarino E., Pastorelli L., Vecchi M., Sconfienza L.M. Bowel Sonoelastography in Patients with Crohn’s Disease: A Systematic Review. Ultrasound Med Biol. 2018 Feb. 44(2). 297-302. doi: 10.1016/j.ultrasmedbio.2017.10.004.
- Greenup A.J., Bressler B., Rosenfeld G. Medical Imaging in Small Bowel Crohn’s Disease-Computer Tomography Enterography, Magnetic Resonance Enterography, and Ultrasound: “Which One Is the Best for What?”. Inflamm Bowel Dis. 2016 May. 22(5). 1246-61. doi: 10.1097/MIB.0000000000000727.
- Baumgart D.C., Müller H.P., Grittner U., Metzke D., Fis–cher A., Guckelberger O., Pascher A. US-based Real-time Elastography for the Detection of Fibrotic Gut Tissue in Patients with Stricturing Crohn Disease. Radiology. 2015 Jun. 275(3). 889-99.
- Dillman J.R., Stidham R.W., Higgins P.D., Moons D.S., Johnson L.A., Rubin J.M. US elastography-derived shear wave velo–city helps distinguish acutely inflamed from fibrotic bowel in a Crohn disease animal model. Radiology. 2013 Jun. 267(3). 757-66.
- Fraquelli M., Branchi F., Cribiù F.M., Orlando S., Casaz–za G., Magarotto A., Massironi S. The Role of Ultrasound Elasticity Imaging in Predicting Ileal Fibrosis in Crohn’s Disease Patients. Inflamm Bowel Dis. 2015 Nov. 21(11). 2605-12.
- Vieujean S., Hu S., Bequet E. et al. Potential Role of Epithelial Endoplasmic Reticulum Stress and Anterior Gradient Protein 2 Homologue in Crohn’s Disease Fibrosis. J Crohns Colitis. 2021 Oct 7. 15(10). 1737-1750. doi: 10.1093/ecco-jcc/jjab061.
- Rieder F., Latella G., Magro F., Yuksel E.S., Higgins P.D., Di Sabatino A., de Bruyn J.R. European Crohn’s and Colitis Organisation Topical Review on Prediction, Diagnosis and Management of Fibrostenosing Crohn’s Disease. J Crohns Colitis. 2016 Aug. 10(8). 873-85.
- Gajendran M., Loganathan P., Catinella A.P., Hashash J.G. A comprehensive review and update on Crohn’s disease. Dis Mon. 2018 Feb. 64(2). 20-57. doi: 10.1016/j.disamonth.2017.07.001.
- Rimola J., Planell N., Rodríguez S., Delgado S., Ordás I., Ramírez-Morros A., Ayuso C., Characterization of inflammation and fibrosis in Crohn’s disease lesions by magnetic resonance imaging. Am J Gastroenterol. 2015 Mar. 110(3). 432-40.
- Bettenworth D., Nowacki T.M., Cordes F., Buerke B., Lenze F. Assessment of stricturing Crohn’s disease: Current clinical practice and future avenues. World J Gastroenterol. 2016 Jan 21. 22(3). 1008-16. doi: 10.3748/wjg.v22.i3.1008.
- Ippolito D., Lombardi S., Talei Franzesi C. et al. Dynamic Contrast-Enhanced MR with Quantitative Perfusion Analysis of Small Bowel in Vascular Assessment between Inflammatory and Fibrotic Lesions in Crohn’s Disease: A Feasibility Study. Contrast Media Mol Imaging. 2019 Feb 4. 2019. 1767620. doi: 10.1155/2019/ 1767620.
- Catalano O.A., Rosen B.R., Sahani D.V., Hahn P.F., Guimaraes A.R., Vangel M.G., Nicolai E. Clinical impact of PET/MR imaging in patients with cancer undergoing same-day PET/CT: initial experience in 134 patients--a hypothesis-generating exploratory study. Radiology. 2013 Dec. 269(3). 857-69.
- Catalano O.A., Gee M.S., Nicolai E., Selvaggi F., Pellino G., Cuocolo A., Luongo A. Evaluation of Quantitative PET/MR Entero–graphy Biomarkers for Discrimination of Inflammatory Strictures from Fibrotic Strictures in Crohn Disease. Radiology. 2016 Mar. 278(3). 792-800.
- Pellino G., Pallante P., Selvaggi F. Novel biomarkers of fibrosis in Crohn’s disease. World J Gastrointest Pathophysiol. 2016 Aug 15. 7(3). 266-75.
- Macedo C.P., Sarmento Costa M., Gravito-Soares E., Gravito-Soares M., Ferreira A.M., Portela F., Figueiredo P. Role of Intestinal Ultrasound in the Evaluation of Postsurgical Recurrence in Crohn’s Disease: Correlation with Endoscopic Findings. GE Port J Gastroenterol. 2021 Aug 12. 29(3). 178-186. doi: 10.1159/000517999.
- Kang E.A., Jang J., Choi C.H. et al. Development of a Clinical and Genetic Prediction Model for Early Intestinal Resection in Patients with Crohn’s Disease: Results from the IMPACT Study. J Clin Med. 2021 Feb 7. 10(4). 633. doi: 10.3390/jcm10040633.
- Gordon I.O., Agrawal N., Willis E. et al. Fibrosis in ulcera–tive colitis is directly linked to severity and chronicity of mucosal inflammation. Aliment Pharmacol Ther. 2018 Apr. 47(7). 922-939. doi: 10.1111/apt.14526.
- Wang J., Lin S., Brown J.M., van Wagoner D., Fiocchi C., Rieder F. Novel mechanisms and clinical trial endpoints in intestinal fibrosis. Immunol Rev. 2021 Jul. 302(1). 211-227. doi: 10.1111/imr.12974.